Breast cancer
Worldwide, breast cancer comprises 10.4% of all cancer incidence among women, making it the second most common type of non-skin cancer (after lung cancer) and the fifth most common cause of cancer death. In 2004, breast cancer caused 519,000 deaths worldwide (7% of cancer deaths; almost 1% of all deaths). Breast cancer is about 100 times more common in women than in men, but survival rates are equal in both sexes.
Some breast cancers require the hormones estrogen and progesterone to grow, and have receptors for those hormones. After surgery those cancers are treated with drugs that interfere with those hormones, usually tamoxifen, and with drugs that shut off the production of estrogen in the ovaries or elsewhere; this may damage the ovaries and end fertility. Low-risk, hormone-sensitive breast cancers may be treated with hormone therapy and radiation alone. Breast cancers without hormone receptors, or which have spread to the lymph nodes in the armpits, or which express certain genetic characteristics, are higher-risk, and are treated more aggressively. One standard regimen, popular in the U.S., is cyclophosphamide plus doxorubicin (Adriamycin), known as CA; these drugs damage DNA in the cancer, but also in fast-growing normal cells where they cause serious side effects. Sometimes a taxane drug, such as docetaxel, is added, and the regime is then known as CAT; taxane attacks the microtubules in cancer cells. An equivalent treatment, popular in Europe, is cyclophosphamide, methotrexate, and fluorouracil (CMF). Monoclonal antibodies, such as trastuzumab, are used for cancer cells that have the HER2 mutation. Radiation is usually added to the surgical bed to control cancer cells that were missed by the surgery, which usually extends survival, although radiation exposure to the heart may cause damage and heart failure in the following years.
Classification
- Stage. The TNM classification for breast cancer is based on the size of the tumor (T), whether or not the tumor has spread to the lymph nodes (N) in the armpits, and whether the tumor has metastasized (M) or spread to a more distant part of the body. Larger size, nodal spread, and metastasis have a worse prognosis.
- Pathology. Most breast cancers are’ derived from the epithelium lining the ducts or lobules. (Cancers from other tissues are considered “rare” cancers.) Carcinoma in situ is proliferation of cancer cells within the epithelial tissue without invasion of the surrounding tissue. Invasive carcinoma invades the surrounding tissue. Cells that are dividing more quickly have a worse prognosis. One way to measure tumor cell growth is with the presence of protein Ki67, which indicates that the cell is in S phase, and also indicates susceptibility to certain treatments.
- Grade (Bloom-Richardson grade). When cells become differentiated, they take different shapes and forms to function as part of an organ. Cancerous cells lose that differentiation. Cells that normally line up in an orderly way to make up the milk ducts become disorganized. Cell division becomes uncontrolled. Cell nuclei become less uniform. Pathologists describe cells as well differentiated (low grade), moderately differentiated (intermediate grade), and poorly differentiated (high grade). Poorly-differentiated cancers have a worse prognosis.
- Receptor status. Breast cancer cells have receptors on their surface. Chemical messengers such as hormones bind to receptors, and this causes changes in the cell. Breast cancer cells may or may not have three important receptors: estrogen receptor (ER), progesterone receptor (PR), and HER2/neu. Cells with these receptors are called ER positive (ER+), ER negative (ER-), PR positive (PR+), PR negative (PR-), HER2 positive (HER2+), and HER2 negative (HER2-). Cells with none of these receptors are called basal-like or triple negative. ER+ cancer cells depend on estrogen for their growth, so they can be treated with drugs to reduce estrogen, and generally have a better prognosis. HER2+ cancer cells respond to drugs such as trastuzumab, and to high doses of doxorubicin. Generally, HER2+ has a worse prognosis. These receptors are identified by immunohistochemistry.
- Receptor status is used to divide breast cancer into four molecular classes: (1) Basal-like, which are ER-, PR- and HER2- (triple negative, TN). Most BRCA1 breast cancers are basal-like TN. (2) Luminal A, which are ER+ and low grade (3) Luminal B, which are ER+ but often high grade (4) HER2+, which have amplified ERBB2.
- DNA microarrays have compared normal cells to breast cancer cells and found differences in hundreds of genes, but the significance of most of those differences is unknown. Several screening tests are commercially marketed, but the evidence for their value is limited. The only test supported by Level II evidence is Oncotype DX, which is not approved by the U.S. Food and Drug Administration (FDA) but is endorsed by the American Society of Clinical Oncology. MammaPrint is approved by the FDA but is only supported by Level III evidence. Two other tests have Level III evidence: Theros and MapQuant Dx. No tests have been verified by Level I evidence (a prospective, randomized controlled trial in which patients who used the test had a better outcome than those who did not). In a review, Sotirou concluded, “The genetic tests add modest prognostic information for patients with HER2-positive and triple-negative tumors, but when measures of clinical risk are equivocal (e.g., intermediate expression of ER and intermediate histologic grade), these assays could guide clinical decisions.”
Breast cancer is usually, but not always, primarily classified by its histological appearance. Rare variants are defined on the basis of physical exam findings. For example, inflammatory breast cancer (IBC), a form of ductal carcinoma or malignant cancer in the ducts, is distinguished from other carcinomas by the inflamed appearance of the affected breast. In the future, some pathologic classifications may be changed.
Signs and symptoms
Indications of breast cancer other than a lump may include changes in breast size or shape, skin dimpling, nipple inversion, or spontaneous single-nipple discharge. Pain (“mastodynia”) is an unreliable tool in determining the presence or absence of breast cancer, but may be indicative of other breast health issues.
When breast cancer cells invade the dermal lymphatics—small lymph vessels in the skin of the breast—its presentation can resemble skin inflammation and thus is known as inflammatory breast cancer (IBC). Symptoms of inflammatory breast cancer include pain, swelling, warmth and redness throughout the breast, as well as an orange-peel texture to the skin referred to as peau d’orange.
Another reported symptom complex of breast cancer is Paget’s disease of the breast. This syndrome presents as eczematoid skin changes such as redness and mild flaking of the nipple skin. As Paget’s advances, symptoms may include tingling, itching, increased sensitivity, burning, and pain. There may also be discharge from the nipple. Approximately half of women diagnosed with Paget’s also have a lump in the breast.
Occasionally, breast cancer presents as metastatic disease, that is, cancer that has spread beyond the original organ. Metastatic breast cancer will cause symptoms that depend on the location of metastasis. Common sites of metastasis include bone, liver, lung and brain. Unexplained weight loss can occasionally herald an occult breast cancer, as can symptoms of fevers or chills. Bone or joint pains can sometimes be manifestations of metastatic breast cancer, as can jaundice or neurological symptoms. These symptoms are “non-specific”, meaning they can also be manifestations of many other illnesses.
Most symptoms of breast disorder do not turn out to represent underlying breast cancer. Benign breast diseases such as mastitis and fibroadenoma of the breast are more common causes of breast disorder symptoms. The appearance of a new symptom should be taken seriously by both patients and their doctors, because of the possibility of an underlying breast cancer at almost any age.
Risk factors
Well established risk factors account for 47% of cases while 5% are attributable to hereditary syndromes. In particular, carriers of the breast cancer susceptibility genes, BRCA1 and BRCA2, are at a 30-40% increased risk for breast and ovarian cancer, depending on in which portion of the protein the mutation occurs.
- Personal history of breast cancer: A woman who had breast cancer in one breast has an increased risk of getting cancer in her other breast.
- Family history: A woman’s risk of breast cancer is higher if her mother, sister, or daughter had breast cancer. The risk is higher if her family member got breast cancer before age 40. Having other relatives with breast cancer (in either her mother’s or father’s family) may also increase a woman’s risk.
- Certain breast changes: Some women have cells in the breast that look abnormal under a microscope. Having certain types of abnormal cells (atypical hyperplasia and lobular carcinoma in situ [LCIS]) increases the risk of breast cancer.
- Race: Breast cancer is diagnosed more often in Caucasian women than Latina, Asian, or African American women.
- No physical activity: Women who are physically inactive throughout life may have an increased risk of breast cancer. Being active may help decrease risk.
Abortion has not been found to be a risk factor for breast cancer. The breast cancer abortion hypothesis however continues to be promoted by some pro-life groups.
United Kingdom being member of International Cancer Genome Consortium is leading efforts to map breast cancer’s complete genome.
Pathophysiology
Overview of signal transduction pathways involved in apoptosis. Mutations leading to loss of apoptosis can lead to tumorigenesis.
Normal cells will commit cell suicide (apoptosis) when they are no longer needed. Until then, they are protected from cell suicide by several protein clusers and pathways. One of the protective pathways is the PI3K/AKT pathway; another is the RAS/MEK/ERK pathway. Sometimes the genes along these protective pathways are mutated in a way that turns them permanently “on”, rendering the cell incapable of committing suicide when it is no longer needed. This is one of the steps that causes cancer in combination with other mutations. Normally, the PTEN protein turns off the PI3K/AKT pathway when the cell is ready for cell suicide. In some breast cancers, the gene for the PTEN protein is mutated, so the PI3K/AKT pathway is stuck in the “on” position, and the cancer cell does not commit suicide.
Mutations that can lead to breast cancer have been experimentally linked to estrogen exposure.
Failure of immune surveillance, a theory in which the immune system removes malignant cells throughout one’s life.
Abnormal growth factor signaling in the interaction between stromal cells and epithelial cells can facilitate malignant cell growth.
People in less-developed countries report lower incidence rates than in developed countries.
In the United States, 10 to 20 percent of patients with breast cancer and patients with ovarian cancer have a first- or second-degree relative with one of these diseases. Mutations in either of two major susceptibility genes, breast cancer susceptibility gene 1 (BRCA1) and breast cancer susceptibility gene 2 (BRCA2), confer a lifetime risk of breast cancer of between 60 and 85 percent and a lifetime risk of ovarian cancer of between 15 and 40 percent. However, mutations in these genes account for only 2 to 3 percent of all breast cancers.
Diagnosis
While screening techniques (which are further discussed below) are useful in determining the possibility of cancer, a further testing is necessary to confirm whether a lump detected on screening is cancer, as opposed to a benign alternative such as a simple cyst.
In a clinical setting, breast cancer is commonly diagnosed using a “triple test” of clinical breast examination (breast examination by a trained medical practitioner), mammography, and fine needle aspiration cytology. Both mammography and clinical breast exam, also used for screening, can indicate an approximate likelihood that a lump is cancer, and may also identify any other lesions. Fine Needle Aspiration and Cytology (FNAC), which may be done in a GP’s office using local anaesthetic if required, involves attempting to extract a small portion of fluid from the lump. Clear fluid makes the lump highly unlikely to be cancerous, but bloody fluid may be sent off for inspection under a microscope for cancerous cells. Together, these three tools can be used to diagnose breast cancer with a good degree of accuracy.
Other options for biopsy include core biopsy, where a section of the breast lump is removed, and an excisional biopsy, where the entire lump is removed.
Screening
Breast cancer screening refers to testing otherwise-healthy women for breast cancer in an attempt to achieve an earlier diagnosis. The assumption is that early detection will improve outcomes. A number of screening test have been employed including: clinical and self breast exams, mammography, genetic screening, ultrasound, and magnetic resonance imaging.
A clinical or self breast exam involves feeling the breast for lumps or other abnormalities. Evidence however does not support its use.[42] Mammographic screening for breast cancer is also controversial. It uses x-rays to examine the breast for any uncharacteristic masses or lumps. The Cochrane collaboration in 2009 concluded that it is unclear whether screening does more good than harm. Many national organizations however recommend it. If mammography is decided upon it should only be done every two years in women between the ages of 50 and 74.
In women at high risk, such as those with a strong family history of cancer, additional testing may include genetic screening and / or magnetic resonance imaging. Genetic screening involves testing for mutations in the BRCA genes.
Treatment
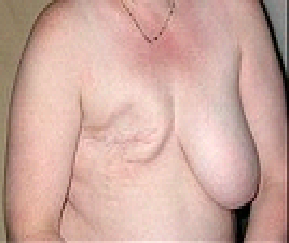
Chest appearance after right breast mastectomy.
Breast cancer is treated first with surgery, and then with drugs, radiation, or both. Treatments are given with increasing aggressiveness according to the prognosis and risk of recurrence. Early cancers with good prognosis (T1, N0) may be treated with lumpectomy plus radiation alone or hormone therapy alone. Later cancers with poorer prognosis and greater risk of recurrence may be treated with more aggressive chemotherapy with uncomfortable and life-threatening side effects, in order to increase the likelihood of cure and lower the risk of recurrence.
Drugs in addition to surgery are called adjuvant therapy. Hormone therapy is one class of adjuvant therapy. Some breast cancers require estrogen to continue growing. They can be identified by the presence of estrogen receptors (ER+) and progesterone receptors (PR+) on their surface (sometimes referred to together as hormone receptors, HR+). These ER+ cancers can be treated with drugs that block the production of estrogen or block the receptors, such as tamoxifen or an aromatase inhibitor).
Chemotherapy is given for more advanced stages of disease. They are usually given in combinations. One of the most common treatments is cyclophosphamide plus doxorubicin (Adriamycin), known as CA; these drugs damage DNA in the cancer, but also in fast-growing normal cells where they cause serious side effects. Damage to the heart muscle is the most dangerous complication of doxorubicin. Sometimes a taxane drug, such as docetaxel, is added, and the regime is then known as CAT; taxane attacks the microtubules in cancer cells. Another common treatment, which produces equivalent results, is cyclophosphamide, methotrexate, and fluorouracil (CMF). (Chemotherapy can literally refer to any drug, but it is usually used to refer to traditional non-hormone treatments for cancer.)
Monoclonal antibodies are sometimes used. Some cancer cells have a receptor called HER2 on their surface. This receptor is normally stimulated by a growth factor which causes the cell to divide; in the absence of the growth factor, the cell normally stops growing. In breast cancer, the HER2 receptor is stuck in the “on” position (constitutively activated). The cell divides without stopping. Trastuzumab, a monoclonal antibody to HER2, can improve survival in these cancers. Other monoclonal antibodies can interrupt other cancer mechanisms in the cell.
Radiotherapy is given after surgery to the region of the tumor bed, to destroy microscopic tumors that may have escaped surgery. Radiation therapy can be delivered as external beam radiotherapy or as brachytherapy (internal radiotherapy). Radiation can reduce the risk of recurrence by 50-66% (1/2 – 2/3rds reduction of risk) when delivered in the correct dose.
Treatments are constantly being evaluated in randomized, controlled trials, to evaluate and compare individual drugs, combinations of drugs, and surgical and radiation techniques. The latest research is reported annually at scientific meetings such as that of the American Society of Clinical Oncology, San Antonio Breast Cancer Symposium, and the St. Gallen Oncology Conference in St. Gallen, Switzerland. These studies are reviewed by professional societies and other organizations, and formulated into guidelines for specific treatment groups and risk category.
Prognosis
A prognosis is a prediction of outcome, usually the probability of death (or survival), and the probability of progression-free survival (PFS) or disease-free survival (DFS). These predictions are based on experience with breast cancer patients with similar classification. A prognosis is an estimate, as patients with the same classification will survive a different amount of time, and classifications are not always precise. Survival is usually calculated as an average number of months (or years) that 50% of patients survive, or the percentage of patients that are alive after 1, 5, 15 and 20 years. Prognosis is important for treatment decisions because patients with a good prognosis are usually offered less invasive treatments, such as lumpectomy and radiation or hormone therapy, while patients with poor prognosis are usually offered more aggressive treatment, such as more extensive mastectomy and one or more chemotherapy drugs.
Prognostic factors include staging, tumor size and location, grade, whether disease is systemic (has metastasized, or traveled to other parts of the body), recurrence of the disease, and age of patient.
Stage is the most important, as it takes into consideration size, local involvement, lymph node status and whether metastatic disease is present. The higher the stage at diagnosis, the worse the prognosis. The stage is raised by the invasiveness of disease to lymph nodes, chest wall, skin or beyond, and the aggressiveness of the cancer cells. The stage is lowered by the presence of cancer-free zones and close-to-normal cell behaviour (grading). Size is not a factor in staging unless the cancer is invasive. Ductal Carcinoma in situ throughout the entire breast is stage zero.
Grading is based on how biopsied, cultured cells behave. The closer to normal cancer cells are, the slower their growth and the better the prognosis. If cells are not well differentiated, they will appear immature, will divide more rapidly, and will tend to spread. Well differentiated is given a grade of 1, moderate is grade 2, while poor or undifferentiated is given a higher grade of 3 or 4 (depending upon the scale used).
Younger women tend to have a poorer prognosis than post-menopausal women due to several factors. Their breasts are active with their cycles, they may be nursing infants, and may be unaware of changes in their breasts. Therefore, younger women are usually at a more advanced stage when diagnosed. There may also be biologic factors contributing to a higher risk of disease recurrence for younger women with breast cancer.
The presence of estrogen and progesterone receptors in the cancer cell, while not prognostic, is important in guiding treatment. Those who do not test positive for these specific receptors will not respond to hormone therapy.
Likewise, HER2/neu status directs the course of treatment. Patients whose cancer cells are positive for HER2/neu have more aggressive disease and may be treated with trastuzumab, a monoclonal antibody that targets this protein.
Elevated CA15-3, in conjunction with alkaline phosphatase, was shown to increase chances of early recurrence in breast cancer.
Psychological aspects
The emotional impact of cancer diagnosis, symptoms, treatment, and related issues can be severe. Most larger hospitals are associated with cancer support groups which provide a supportive environment to help patients cope and gain perspective from cancer survivors. Online cancer support groups are also very beneficial to cancer patients, especially in dealing with uncertainty and body-image problems inherent in cancer treatment.
Not all breast cancer patients experience their illness in the same manner. Factors such as age can have a significant impact on the way a patient copes with a breast cancer diagnosis. Premenopausal women with estrogen-receptor positive breast cancer must confront the issues of early menopause induced by many of the chemotherapy regimens used to treat their breast cancer, especially those that use hormones to counteract ovarian function.
On the other hand, a recent study conducted by researchers at the College of Public Health of the University of Georgia showed that older women may face a more difficult recovery from breast cancer than their younger counterparts. As the incidence of breast cancer in women over 50 rises and survival rates increase, breast cancer is increasingly becoming a geriatric issue that warrants both further research and the expansion of specialized cancer support services tailored for specific age groups.